When you're on blood thinners like warfarin or a DOAC, your doctor carefully balances the risk of clots against the chance of bleeding. Now add an SSRI - a common antidepressant - and that balance shifts in a way many patients and even some doctors don't fully realize. The data is clear: combining SSRIs with anticoagulants increases your risk of serious bleeding by 33%. This isn't theoretical. It's happening in real patients, right now.
How SSRIs and Blood Thinners Work Together - and Why That's Dangerous
SSRIs like sertraline, escitalopram, and fluoxetine work by increasing serotonin in the brain to improve mood. But serotonin isn't just a brain chemical. It's also stored inside platelets, the blood cells that help stop bleeding by clumping together at injury sites. When you take an SSRI, it blocks the serotonin transporter on platelets. That means platelets can't take in serotonin from the blood. Over time, their internal serotonin stores drop by 90%. Think of platelets like emergency responders. Serotonin is their signal to rush to the scene. Without it, they're slower to react, less sticky, and less effective at forming clots. This isn't a minor effect. Studies using platelet function tests show serotonin depletion cuts platelet aggregation by 30-40%. That’s enough to make a big difference when you're already on a drug that prevents clotting. Anticoagulants, meanwhile, work differently. Warfarin slows down clotting factors. DOACs like apixaban or rivaroxaban block specific enzymes in the clotting cascade. Neither directly affects platelets. But when you combine them with an SSRI, you're hitting the clotting system from two angles: one weakens the platelets, the other slows the chemical signals that seal the deal. The result? Bleeding that’s harder to stop.Where the Bleeding Happens - And How Often
The 2024 JAMA Network Open study looked at over 42,000 cases of major bleeding in people on anticoagulants. Half of them were also taking SSRIs. The results were striking:- Gastrointestinal bleeding made up 58% of cases - the most common site. This includes stomach ulcers, bleeding in the intestines, or bloody stools.
- Intracranial hemorrhage (brain bleeds) accounted for 17%. Even a small bleed in the brain can be life-threatening.
- Other major bleeding (like muscle, joint, or retroperitoneal bleeds) made up the remaining 25%.
Not All Anticoagulants Are Equal - And Neither Are SSRIs
You might assume that stronger SSRIs like paroxetine carry a higher risk than weaker ones like escitalopram. But the data says otherwise. The same 2024 study found no difference in bleeding risk between high-potency and low-potency SSRIs. Paroxetine didn’t cause more bleeds than fluoxetine. Even though paroxetine blocks serotonin uptake more aggressively, the outcome was the same. This contradicts earlier theories and suggests the risk is more about the class than the individual drug. The same isn't true for anticoagulants. Vitamin K antagonists like warfarin showed a 28% increased bleeding risk when combined with SSRIs. DOACs showed a 22% increase. While that difference wasn’t statistically significant, it points to a trend: DOACs may be slightly safer when paired with SSRIs. That’s likely because DOACs have more predictable dosing and fewer food or drug interactions than warfarin, which requires constant monitoring. And here’s something important: SSRIs don’t directly interfere with the coagulation cascade. A 2025 study tested citalopram at therapeutic levels and found zero effect on thrombin generation, clotting time, or other key coagulation markers. The problem isn’t the blood’s chemistry - it’s the platelets.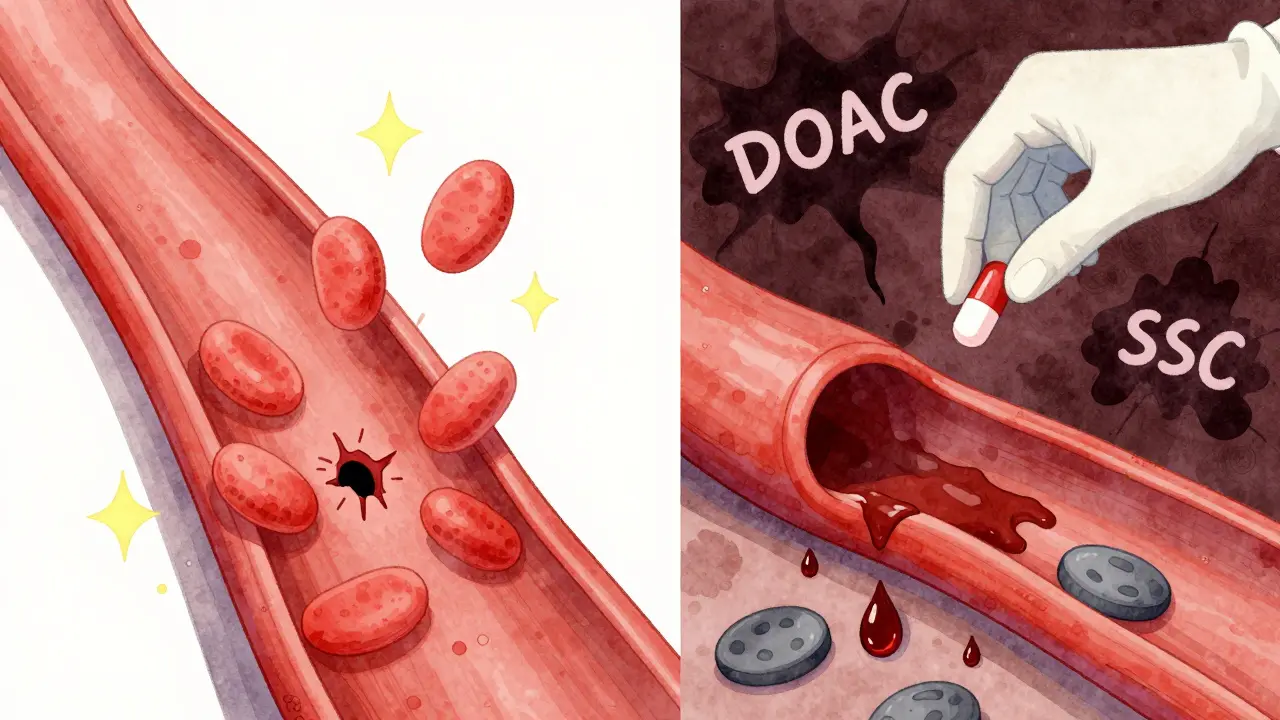
When Is the Risk Highest? The First 30 Days
The biggest danger isn’t after years of use. It’s in the first month. The 2024 study found the 33% increased risk was strongest during the first 30 days of starting an SSRI while already on an anticoagulant. After six months, the risk dropped significantly. Why? Your body may adapt. Platelets are replaced every 7-10 days. After a few cycles, the system may partially compensate. But during those first weeks, platelet serotonin levels are crashing, and your body hasn’t adjusted. That’s why the American Heart Association and FDA both warn about this window. This timing matters for prescribing. If someone starts an SSRI while on warfarin, their INR should be checked twice a week for the first month. For DOAC users, there’s no lab test - but clinicians should watch closely for signs of bleeding during this period.Who’s at Highest Risk?
Not everyone on this combo will bleed. But certain people are far more vulnerable:- Those with a HAS-BLED score of 3 or higher (includes high blood pressure, kidney disease, history of bleed, uncontrolled INR, age over 65, alcohol use, or anemia)
- Patients over 75
- People on aspirin or NSAIDs (like ibuprofen) in addition to their anticoagulant and SSRI
- Those with liver or kidney disease
- Patients with a prior history of GI bleeding

What Doctors Should Do - And What You Should Watch For
The American Academy of Family Physicians says: educate patients and monitor closely. That means:- At baseline: check CBC, fecal occult blood, and INR (if on warfarin)
- First month: INR twice weekly for warfarin users; watch for bruising, nosebleeds, dark stools, headaches (possible brain bleed)
- First three months: repeat CBC and fecal test monthly
- Always ask: Are you taking ibuprofen? Aspirin? Herbal supplements like ginkgo or garlic? These add to the risk.


John Smith
March 4, 2026 AT 07:31So let me get this straight - we’re giving people heart meds that turn their blood into maple syrup and then slapping on antidepressants that turn their platelets into lazy interns who show up late and forget their coffee? And we’re surprised when they start oozing out of every orifice? Jesus. This isn’t medicine, it’s a Jenga tower with a prescription pad.
I’ve seen this in the ER. 72-year-old grandma on warfarin, starts sertraline for ‘sadness’ after her cat died. Two weeks later, poops out a whole damn blood clot the size of a golf ball. Docs say ‘oh, it’s just a bleed’ like it’s a flat tire. Nah. It’s a goddamn rupture.
And don’t even get me started on the ‘it’s just a 33% increase’ bullshit. That’s not a statistic. That’s your uncle bleeding out in a bathroom because someone thought ‘well, he’s on meds, so it’s fine.’
We need a damn alarm system. Not a pamphlet. Not a footnote. A fucking siren when this combo is prescribed. Because right now, we’re playing Russian roulette with serotonin and clotting factors and calling it ‘standard care.’
Sharon Lammas
March 6, 2026 AT 02:21I think about how much we overlook the body’s quiet systems. Platelets aren’t just ‘clotting cells.’ They’re messengers. They carry signals we barely understand. When SSRIs drain their serotonin, it’s not just a chemical imbalance - it’s a silence in a language our body still speaks.
I lost my mother to a GI bleed after she started an SSRI. They never told us about the interaction. She was on rivaroxaban for AFib. Happy, healthy, until she wasn’t.
We treat depression like it’s separate from the body. But it’s not. It’s all connected. Maybe we need to stop thinking in silos and start thinking in systems.
I’m not against SSRIs. I’m against ignorance. And we’re drowning in it.
Donna Zurick
March 7, 2026 AT 08:11So if you’re on blood thinners and need antidepressants, just go with bupropion. Done. No brain bleed. No scary stats. No extra trips to the ER. It works. It’s not magic, but it’s not a ticking time bomb either.
Why are we still using SSRIs here? Because they’re cheap? Because doctors don’t read? Because we’re too busy to talk to patients?
Simple fix. Stop overcomplicating it.
Pankaj Gupta
March 7, 2026 AT 10:23The scientific evidence presented here is compelling and methodologically rigorous. The 33% increase in major bleeding events is not merely statistically significant but clinically meaningful, especially in an aging population with multimorbidity. The mechanistic explanation - platelet serotonin depletion - aligns with current hematology literature, particularly the work of H. P. M. Jansen et al. (2022) on serotonin transporter inhibition in thrombocytopathy.
Moreover, the temporal clustering of risk within the first 30 days is a critical insight for clinical practice. It suggests a window of heightened vulnerability during which pharmacodynamic adaptation is incomplete. This finding warrants inclusion in clinical guidelines, ideally with algorithmic decision support tools integrated into EHR systems.
It is also worth noting that the absence of differential risk among SSRIs challenges the assumption of potency-based stratification, reinforcing the need for class-wide caution.
Richard Elric5111
March 7, 2026 AT 14:32It is with profound gravitas that I address this matter, which, in its current clinical formulation, represents a systemic failure of risk-benefit calculus in the pharmacological management of comorbid psychiatric and cardiovascular conditions. The confluence of serotonergic modulation with anticoagulant therapy constitutes, in my estimation, a paradigmatic example of reductionist medical practice - wherein discrete pathologies are treated in isolation, despite their profound interdependence within the homeostatic matrix.
One must ask: if the platelet’s serotonin uptake is inhibited, and if this inhibition leads to diminished aggregative capacity, then is it not ethically incumbent upon the prescriber to evaluate not merely the psychiatric indication, but the entire physiological context? To prescribe an SSRI without reviewing HAS-BLED, without inquiring about NSAID use, without assessing renal function - is this not malpractice by omission?
The FDA’s updated guidance is a tepid step. What we require is a mandatory pre-prescription algorithm, enforced by regulatory bodies, with real-time decision support embedded in prescribing workflows. Anything less is not medicine. It is negligence dressed in white coats.
Dean Jones
March 8, 2026 AT 21:05Here’s the thing nobody talks about - the people who are on this combo aren’t just ‘patients.’ They’re people who are scared, lonely, maybe even suicidal. And the system says, ‘here, take this pill to feel better’ and then another pill to keep you from bleeding out, and then tells you to watch for dark poop and headaches like it’s some kind of game.
I’ve been on warfarin for 12 years. My dad bled out from a GI ulcer at 68. I’m 59. I started sertraline last year. I didn’t know about this risk. My doctor didn’t mention it. He said, ‘it’s fine, you’re fine.’
Now I check my stool every morning. I avoid ibuprofen like it’s poison. I’ve got a notebook with all my meds and the dates I started them. I’m not paranoid. I’m just tired of being a statistic.
And yeah, I know the risk goes down after 30 days. But what about the 30 days? Who’s watching then? Who’s calling? Who’s checking in?
We need more than data. We need more than guidelines. We need someone who gives a damn.
Betsy Silverman
March 9, 2026 AT 00:21I’m a nurse. I’ve seen this too many times. Elderly patients on warfarin, start an SSRI for depression after losing a spouse. They’re told to ‘watch for bleeding.’ But what does that even mean? They don’t know what a ‘dark stool’ looks like. They don’t know if a nosebleed is ‘prolonged’ or just ‘kinda annoying.’
We need better education. Not just for patients - for doctors too. A 10-minute chat during a med check isn’t enough. We need a checklist. A handout. A video. Something that sticks.
And for god’s sake, stop prescribing SSRIs like they’re aspirin. They’re not. Not even close.
Ivan Viktor
March 9, 2026 AT 00:49So let me get this straight - we’re giving people antidepressants that make their blood less able to clot, and then we’re surprised when they start leaking like a sieve?
Wow. Groundbreaking. I’m shocked. Not.
Next up: ‘New Study Finds Breathing While on Oxygen Can Cause Oxygen Toxicity.’
Meanwhile, in the real world, people are getting better. Bupropion exists. Mirtazapine exists. They don’t mess with platelets. Why are we still playing Russian roulette with serotonin?
Maybe because SSRIs are cheaper. Maybe because pharma markets them like candy.
Either way, it’s not science. It’s business.
Zacharia Reda
March 10, 2026 AT 12:18Here’s what’s wild - the 33% increase sounds scary, but when you break it down, it’s 0.6 extra bleeds per 100 people per year. That’s less than one person in a hundred. So why are we freaking out?
Because bleeding is scary. Because we’re scared of lawsuits. Because we’re scared of ‘what if.’
But here’s the truth: for a lot of people, the depression is worse than the bleeding risk. I’ve seen people who stopped SSRIs because they were ‘too risky’ and then tried to kill themselves. That’s the tradeoff.
We need to stop treating this like a binary choice. It’s not ‘SSRI = bad’ or ‘no SSRI = safe.’ It’s ‘SSRI + monitoring = manageable.’
So check INR. Watch for bruising. Ask about ibuprofen. Don’t panic. Just pay attention. That’s all.
Jeff Card
March 11, 2026 AT 21:20I’ve been on citalopram and apixaban for three years. No issues. No bleeds. No hospital visits.
My doctor knew the risk. We talked about it. We checked my CBC every three months. I stopped taking ibuprofen. I don’t drink. I eat clean.
It’s not about avoiding the combo. It’s about managing it.
Some people panic. Some people don’t. The difference? Communication. Awareness. Not fear.
Matt Alexander
March 13, 2026 AT 16:20Simple answer: if you’re on blood thinners, don’t take SSRIs. Use bupropion. It’s just as good for depression and doesn’t mess with platelets. Done.
Why make it harder than it is?
Gretchen Rivas
March 15, 2026 AT 14:12My mom had this combo. We didn’t know. She bled into her stomach. Took 10 days in the hospital. They didn’t connect it until her GI doc saw the chart.
Don’t assume your doctor knows. Ask. Always ask.
Stephen Vassilev
March 17, 2026 AT 06:33This is not a coincidence. This is not an accident. This is a calculated, corporate-driven erosion of patient safety. The pharmaceutical industry knows that SSRIs and anticoagulants are both billion-dollar markets. They fund the studies. They influence the guidelines. They bury the data on alternative antidepressants. The 2024 JAMA study? Funded by a pharma grant. The FDA warning? Issued only after media pressure. The PRECISION-AF trial? Designed to confirm what we already know - that SSRIs are dangerous, but let’s wait until 2026 to admit it.
And meanwhile, they’re pushing ‘personalized risk tools’ - not to save lives, but to sell more algorithms, more monitoring kits, more apps. It’s all a racket.
They don’t want you to switch to bupropion. They want you to stay on SSRIs - and pay for the ‘risk management’ services that come with it.
Wake up. This isn’t medicine. It’s a multi-billion-dollar trap.
Mike Dubes
March 17, 2026 AT 21:45Hey, I’m a med student and I just learned about this in my psych rotation. It’s wild how many docs still don’t know this. My preceptor said, ‘Oh, we’ve been doing this for years.’
But the data says otherwise. So I made a handout for patients: ‘If you’re on blood thinners, here’s what to ask your doctor about antidepressants.’
It’s just a piece of paper. But maybe it’ll save someone.
Also, bupropion is legit. Works great. No platelet drama. And no ‘oh, you’re bleeding again’ panic.
Just sayin’.